The power of regeneration
The wide-ranging application of platelet rich growth factor Endoret (PRGF) in surgical dentistry is helping to make outcomes safer and more predictable
“in this simple case, all three forms of Endoret (PRGF) were used to achieve a small lateral augmentation of the implant site “
Due to its powerful regenerative properties, platelet rich growth factor Endoret (PRGF) is being increasingly used in many fields of medicine, such as plastic surgery and dental implantology.
It has been shown to reduce scarring, rejuvenate damaged facial tissue, and accelerate wound healing. Endoret (PRGF) is proven to promote angiogenesis, cell migration, cell proliferation and the secretion of growth factors active in the wound healing cascade, while at the same time decreasing inflammation and pain.
Through this simple case report, I would like to show how Endoret (PRGF) can be applied in the field of dental implantology to make treatment outcomes more predicable by optimising healing conditions.
Preparation of platelet rich growth factor (Endoret)
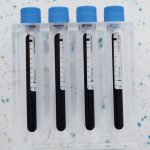
Step one is to collect a small quantity of the patient’s blood. Four collection tubes, each containing 9ml of blood, are filled. A total of 36ml is usually sufficient for most implant cases (Fig 1) However, in larger cases where more augmentation is required it is usual to collect eight tubes (72ml).
Once the blood has been collected, the patient is asked to return to the waiting area while the fractionation process is carried out which takes about 15 minutes. The collection tubes are transferred to the centrifuge machine (Fig 2). At the end of the eight-minute cycle the collection tubes are immediately returned to their stand. It is important that this is done carefully with minimal disruption to the blood, which is now separated into four distinctive bands.
Each tube is now marked. The erythrocytes are heaviest and lie in the bottom half.
Next is a thin buffy layer approximately 0.5cm thick. This is the leukocyte fraction which must be avoided as these cells will evoke pain and inflammation.
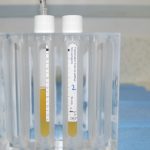
In the top half of the tube is the liquid fraction of interest. Concentrated in platelets, plasma and growth factors, this straw-coloured layer is itself divided into two fractions: 1 and 2.
While fraction 1 (F1) is richer in the fibrin, which will create the collagen matrix for wound healing, fraction 2 (F2) contains the greatest concentration of platelets and protein markers. It is these protein markers which signal to the regenerative cells and trigger their activity.
Marking each tube, a safe distance of 0.5cm from the visible upper limit of the buffy layer, you then measure two centimetres up and mark the tube again.
This threshold divides the most superior layer F1 from the second layer F2 Fig 3.
Using the plasma transfer device, F1 and F2 are then separated into labelled collection tubes F1 and F2. Without activation, these fractions will remain viable for up to four hours.
At this point, I will normally ask for the patient to return and we follow the usual asepsis protocol for surgery and deliver the local anaesthetic and or intravenous sedation where required. When placing implants, I always require two dental surgery assistants, one sterile and the other non-sterile.
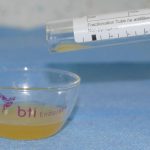
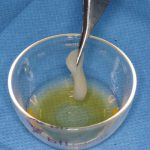
Depending on the planned length of the procedure, I will indicate to my assistants when F1 and F2 should be activated with calcium chloride (Fig 4). Once activated the fractions are transferred into sterile glass bowls and placed into a special oven that incubates them at body temperature for 15-25 minutes (Fig 5).
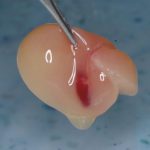
Endoret (PGRF) is used in three distinct forms. There is the clot (F2), into which autogenous bone collected during implant site preparation is added, and also xenograft in situations where more augmentation is required (Fig 6).
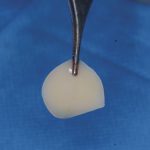
There is the fibrin membrane (F1), which is placed on top of F2 (Fig 7). This stimulates fibroblasts which accelerates wound closure and often creates thicker mucosal biotype.
The liquid form of Endoret (PRGF) is used to promote healing after ridge-splitting procedures, and to improve implant integration by up to 40 per cent when the implant surface is coated immediately before placement.
In this simple case, all three forms of Endoret (PRGF) were used to achieve a small lateral augmentation of the implant site, as well as promote osseointregration of the implant and wound closure by primary intention.
A number 15 scalpel was used to make a palatally inclined incision at the site of the missing upper right first premolar. This cut was extended mesially and distally around the adjacent teeth without relieving incisions.
A number 12 scalpel was then used to make a periosteal releasing incision in the pocket flap created being mindful of the proximity of the infra-orbital nerve.
After the creation of the pilot hole using a drill with irrigation at 1800rpm, the remainder of the osteotomy was created following a biological drilling protocol advocated by Professor Eduardo Anitua, Scientific Director of BTI Biotechnology Institute.
This involves preparation without irrigation at speeds of between 50 and 150 rpm. A high level of control can be maintained at these low speeds while biologically viable autogenous bone debris can be collected from the drill flutes which is transferred to the F2 for later augmentation.
Immediately before implant placement, the selected implant’s entire surface was dipped in
liquid Endoret (PRGF). The liquid was also injected into the osteotomy. Engine placement of the Implant then proceeded as per normal protocol.
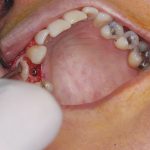
Lateral augmentation was completed easily and safely without the need for a membrane and in this case, without xenograft either. The F2 clot containing the collected autogenous bone drill debris was first placed against the bone (Fig 8). In situations where more lost bone volume needs to be replaced, a second clot of F2 containing xenograft is layered on top of the autogenous layer. Finally, the F1 fibrin membrane was placed on top (Fig 9).
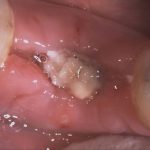
The wound was closed with three interrupted sutures,which were removed after four days (Fig 10).
The autologous nature of Endoret (PRGF) means it has many applications in dentistry beyond the simple implant case described here.
In the atrophic maxilla where residual bone height below the sinus is very low, Endoret (PRGF) in combination with short implants can be used in a transalveolar elevation approach to gain 2-3mm of additional bone height very safely. Such an approach is now being used widely to avoid more traditional and invasive methods such the lateral window technique.
In the field of oral surgery accelerated and improved healing has obvious benefits; for example, the avoidance of dry socket through socket preservation with Endoret (PRGF), and the treatment of bisphosphonate osteoradionecrosis where necrotic bone has been resected.
Combining Endoret (PRGF) with traditional guided bone regeneration procedures significantly reduces the incidence of wound dehiscence as well as eliminating the need for costly and technique sensitive collagen membranes. When mixed with xenografts such as Bio Oss, Endoret (PRGF) will attract the osteogenic cells necessary to promote true bone formation.
Backed up by more than 15 years of research, and with more than 700,000 patients treated
from 20 countries without adverse effects being reported, the applications for Endoret (PRGF) in surgical dentistry are wide-ranging, predictable and safe.
- Fig 1: Collection tubes each containing 9ml of blood
- Fig 2: Centrifuge machine being loaded
- Fig 3: Blood now separated
- Fig 4: Fraction 1 and Fraction 2 collection tubes into fractions
- Fig 5: Fraction 2 being transferred to glass bowl ready for incubation
- Fig 6: Fraction 2 clot containing autogenous bone debris
- Fig 7: Fraction 1 clot shaped in a special press to form a membrane.
- Fig 8: Fraction 2 clot being placed against the bone
- Fig 9: Fraction 1 fibrin membrane positioned over Faction 2 clot
- Fig 10: Healing at four days.
For more information please contact info@gracefielddental.ie
 About the author
About the author
Dr Jerome P Sullivan BDS, DipImpDent RCS (Eng), PG cert sed UCL qualified from Manchester University in 1993. He graduated with
a diploma in dental implantology from the Royal College of Surgeons (UK) in 2011, and was awarded an inaugural gold medal for the high standard of his clinical cases. He has become increasingly involved in the field of platelet rich growth factor, and he now uses this technique to deliver implant solutions
to patients with complex dental needs
Tags: endoret, platlet, regeneration, surgery


You must be logged in to post a comment.